A Data-driven Framework for Modeling Environmental Exposure Mixtures, Biological Aging Acceleration, and Chronic Disease Risk in U.S. Adults

Article Information
Abstract
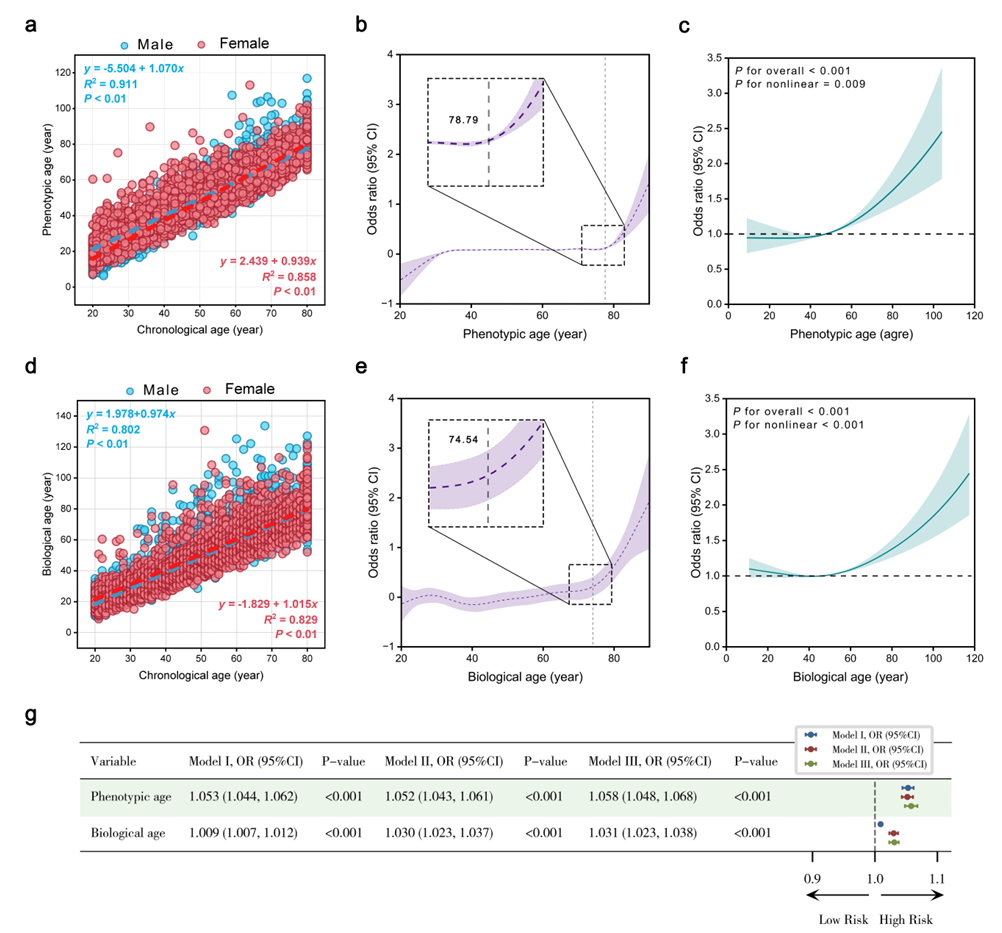
Environmental exposure biomarkers (EEBs) reflect the internal burden of pollutants, yet the joint effects of multiple exposures on biological aging and chronic disease risk remain insufficiently characterized. We analyzed 8,582 adults from the 2013-2016 National Health and Nutrition Examination Survey (NHANES). Mixed exposure was characterized using 74 EEBs. Phenotypic age acceleration and biological age acceleration were used as aging outcomes. Weighted quantile sum (WQS) regression, Bayesian kernel machine regression (BKMR), and LASSO regression were applied to identify key exposure components associated with aging acceleration. Logistic and Cox regression models were then used to evaluate the associations between aging indicators and chronic disease risks. Higher mixed EEB exposure was significantly associated with accelerated aging, reflected by increases in both phenotypic age and biological age. WQS models identified arsenobetaine, copper, and tin as major contributors to phenotypic age acceleration, whereas selenium, zinc, and MHNCH contributed most strongly to biological age acceleration. Moreover, each one-year increase in phenotypic age was associated with a 30.0\% higher risk of dyslipidemia and a 14.3\% higher risk of metabolic-associated fatty liver disease, while each one-year increase in biological age was associated with a 59.7\% higher risk of chronic obstructive pulmonary disease and a 48.5\% higher risk of anemia. This study proposes a unified data-driven analytical framework that integrates exposure mixtures, biological aging, and disease risk modeling. The findings highlight the importance of evaluating mixed exposures rather than single pollutants and may support risk stratification and prevention strategies in environmental health.
Graphical Abstract
Keywords
Data Availability Statement
Funding
Conflicts of Interest
AI Use Statement
Ethical Approval and Consent to Participate
References
- López-Otín, C., Blasco, M. A., Partridge, L., Serrano, M., & Kroemer, G. (2013). The hallmarks of aging. Cell, 153(6), 1194-1217.
[CrossRef] [Google Scholar] - Li, X., & Ma, L. (2024). From biological aging to functional decline: Insights into chronic inflammation and intrinsic capacity. Ageing Research Reviews, 93, 102175.
[CrossRef] [Google Scholar] - Kirkwood, T. B. L., & Austad, S. N. (2000). Why do we age? Nature, 408(6809), 233-238.
[CrossRef] [Google Scholar] - Horvath, S., & Raj, K. (2018). DNA methylation-based biomarkers and the epigenetic clock theory of ageing. Nature reviews genetics, 19(6), 371-384.
[CrossRef] [Google Scholar] - Sorrentino, J. A., Sanoff, H. K., & Sharpless, N. E. (2014). Defining the toxicology of aging. Trends in Molecular Medicine, 20(7), 375-384.
[CrossRef] [Google Scholar] - Belsky, D. W., & Baccarelli, A. A. (2023). To promote healthy aging, focus on the environment. Nature Aging, 3(11), 1334-1344.
[CrossRef] [Google Scholar] - Wahl, H.-W., Iwarsson, S., & Oswald, F. (2012). Aging well and the environment: Toward an integrative model and research agenda for the future. The Gerontologist, 52(3), 306-316.
[CrossRef] [Google Scholar] - Kim, E. S., Tkatch, R., Martin, D., MacLeod, S., Sandy, L., & Yeh, C. (2021). Resilient aging: Psychological well-being and social well-being as targets for the promotion of healthy aging. Gerontology and Geriatric Medicine, 7, 23337214211002951.
[CrossRef] [Google Scholar] - Ferrucci, L., Levine, M. E., Kuo, P.-L., & Simonsick, E. M. (2018). Time and the metrics of aging. Circulation Research, 123(7), 740-744.
[CrossRef] [Google Scholar] - Wu, H., Eckhardt, C. M., & Baccarelli, A. A. (2023). Molecular mechanisms of environmental exposures and human disease. Nature Reviews Genetics, 24(5), 332-344.
[CrossRef] [Google Scholar] - Wang, C., Koutrakis, P., Gao, X., Baccarelli, A., & Schwartz, J. (2020). Associations of annual ambient PM2.5 components with DNAm PhenoAge acceleration in elderly men: The Normative Aging Study. Environmental Pollution, 258, 113690.
[CrossRef] [Google Scholar] - Nwanaji-Enwerem, J. C., Van Ngu, T., Cardenas, A., Baccarelli, A. A., & Nunez-Smith, M. (2021). Adopting a “compound” exposome approach in environmental aging biomarker research: A call to action for advancing racial health equity. Environmental Health Perspectives, 129(4), 045001.
[CrossRef] [Google Scholar] - Wang, S., Ma, N., Kawanishi, S., Hiraku, Y., Oikawa, S., Xie, Y., Zhang, Z., Huang, G., & Murata, M. (2024). Particulate matter 2.5 accelerates aging: Exploring cellular senescence and age-related diseases. Ecotoxicology and Environmental Safety, 284, 116920.
[CrossRef] [Google Scholar] - Davalli, P., Mitic, T., Caporali, A., Lauriola, A., & D'Arca, D. (2016). ROS, cell senescence, and novel molecular mechanisms in aging and age-related diseases. Oxidative Medicine and Cellular Longevity, 2016(1), 3565127.
[CrossRef] [Google Scholar] - Rottenberg, H., & Hoek, J. B. (2017). The path from mitochondrial ROS to aging runs through the mitochondrial permeability transition pore. Aging Cell, 16(5), 943-955.
[CrossRef] [Google Scholar] - Santos, A. L., Sinha, S., & Lindner, A. B. (2018). The good, the bad, and the ugly of ROS: New insights on aging and aging-related diseases from eukaryotic and prokaryotic model organisms. Oxidative Medicine and Cellular Longevity, 2018(1), 1941285.
[CrossRef] [Google Scholar] - Andrade, B., Jara-Gutiérrez, C., Paz-Araos, M., Vásquez-Trincado, C., & Radojkovic, C. (2022). The relationship between reactive oxygen species and the cGAS/STING signaling pathway in the inflammaging process. International Journal of Molecular Sciences, 23(23), 15182.
[CrossRef] [Google Scholar] - Guachalla, L. M., & Rudolph, K. L. (2010). ROS induced DNA damage and checkpoint responses: Influences on aging? Cell Cycle, 9(20), 4058-4060.
[CrossRef] [Google Scholar] - Afanas’ev, I. (2013). New nucleophilic mechanisms of ROS-dependent epigenetic modifications: Comparison of aging and cancer. Aging and Disease, 5(1), 52.
[CrossRef] [Google Scholar] - Wu, Z., Du, Y., Xue, H., Wu, Y., & Zhou, B. (2012). Aluminum induces neurodegeneration and its toxicity arises from increased iron accumulation and reactive oxygen species (ROS) production. Neurobiology of Aging, 33(1), 199.e1-199.e12.
[CrossRef] [Google Scholar] - MacNee, W. (2016). Is chronic obstructive pulmonary disease an accelerated aging disease? Annals of the American Thoracic Society, 13(Supplement 5), S429-S437.
[CrossRef] [Google Scholar] - Blackburn, E. H., Epel, E. S., & Lin, J. (2015). Human telomere biology: A contributory and interactive factor in aging, disease risks, and protection. Science, 350(6265), 1193-1198.
[CrossRef] [Google Scholar] - Guo, J., Huang, X., Dou, L., Yan, M., Shen, T., Tang, W., & Li, J. (2022). Aging and aging-related diseases: From molecular mechanisms to interventions and treatments. Signal Transduction and Targeted Therapy, 7(1), 391.
[CrossRef] [Google Scholar] - Cao, Q., Wu, S., & Xiao, T. (2024). Biological aging mediates the association between volatile organic compounds and cardiovascular disease. BMC Public Health, 24(1), 2928.
[CrossRef] [Google Scholar] - Cakmak, S., Cole, C., Hebbern, C., Andrade, J., & Dales, R. (2020). Associations between blood volatile organic compounds, and changes in hematologic and biochemical profiles, in a population-based study. Environment International, 145, 106121.
[CrossRef] [Google Scholar] - Ogbodo, J. O., Arazu, A. V., Iguh, T. C., Onwodi, N. J., & Ezike, T. C. (2022). Volatile organic compounds: A proinflammatory activator in autoimmune diseases. Frontiers in Immunology, 13, 928379.
[CrossRef] [Google Scholar] - Chen, X., Ren, H., Wang, C., & Zhang, J. (2024). Associations between ethylene oxide exposure and biological age acceleration: Evidence from NHANES 2013–2016. Frontiers in Public Health, 12, 1488558.
[CrossRef] [Google Scholar] - Fu, Z., Liu, Y., & Xu, Q. (2024). Association between single and mixed exposure to polycyclic aromatic hydrocarbons and biological aging. Frontiers in Public Health, 12, 1379252.
[CrossRef] [Google Scholar] - Yang, G., Weng, Z., & Chen, X. (2025). Biological age acceleration and interaction with genetic predisposition in the risk of type 2 diabetes and coronary artery disease. Geroscience, 1-16.
[CrossRef] [Google Scholar] - Ahluwalia, N., Dwyer, J., Terry, A., Moshfegh, A., & Johnson, C. (2016). Update on NHANES dietary data: Focus on collection, release, analytical considerations, and uses to inform public policy. Advances in Nutrition, 7(1), 121-134.
[CrossRef] [Google Scholar] - Yao, X., Xu, X., Wang, G., Lei, M., Qu, L., & Huo, X. (2021). Stratification of population in NHANES 2009-2014 based on exposure pattern of lead, cadmium, mercury, and arsenic and their association with cardiovascular, renal and respiratory outcomes. Environment International, 149, 106410.
[CrossRef] [Google Scholar] - Park, H. S., & Jun, C. H. (2009). A simple and fast algorithm for K-medoids clustering. Expert systems with applications, 36(2), 3336-3341.
[CrossRef] [Google Scholar] - World Medical Association. (2025). World Medical Association Declaration of Helsinki: Ethical principles for medical research involving human participants. JAMA, 333(1), 71-74.
[CrossRef] [Google Scholar] - Dong, H., Yang, X., Zhang, S., Wang, X., Guo, C., Zhang, X., ... & Chen, T. (2021). Associations of low level of fluoride exposure with dental fluorosis among US children and adolescents, NHANES 2015–2016. Ecotoxicology and environmental safety, 221, 112439.
[CrossRef] [Google Scholar] - Chobanian, A. V., Bakris, G. L., Black, H. R., Cushman, W. C., Green, L. A., Izzo Jr, J. L., ... & National High Blood Pressure Education Program Coordinating Committee, T. (2003). The seventh report of the joint national committee on prevention, detection, evaluation, and treatment of high blood pressure: the JNC 7 report. Jama, 289(19), 2560-2571.
[CrossRef] [Google Scholar] - Dang, K., Wang, X., Hu, J., Zhang, Y., Cheng, L., Qi, X., ... & Li, Y. (2024). The association between triglyceride-glucose index and its combination with obesity indicators and cardiovascular disease: NHANES 2003–2018. Cardiovascular diabetology, 23(1), 8.
[CrossRef] [Google Scholar] - Ba, Y., Yu, H., Wu, F., Wang, L., & Ren, X. (2023). Association of exposures to serum terpenes with the prevalence of dyslipidemia: A population-based analysis. Environmental Science and Pollution Research, 30(54), 115295-115309.
[CrossRef] [Google Scholar] - Mach, F., Baigent, C., Catapano, A. L., Koskinas, K. C., Casula, M., Badimon, L., ... & Wiklund, O. (2020). 2019 ESC/EAS Guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk: the Task Force for the management of dyslipidaemias of the European Society of Cardiology (ESC) and European Atherosclerosis Society (EAS). European heart journal, 41(1), 111-188.
[CrossRef] [Google Scholar] - Langsted, A., Madsen, C. M., & Nordestgaard, B. G. (2020). Contribution of remnant cholesterol to cardiovascular risk. Journal of Internal Medicine, 288(1), 116-127.
[CrossRef] [Google Scholar] - Wang, Y. F., Xie, B., & Zou, Y. X. (2023). Association between PFAS congeners exposure and asthma among US children in a nationally representative sample. Environmental Geochemistry and Health, 45(8), 5981-5990.
[CrossRef] [Google Scholar] - Rahman, H. H., Niemann, D., & Munson-McGee, S. H. (2022). Association between environmental toxic metals, arsenic and polycyclic aromatic hydrocarbons and chronic obstructive pulmonary disease in the US adult population. Environmental Science and Pollution Research, 29(36), 54507-54517.
[CrossRef] [Google Scholar] - Shiue, I. (2016). Urinary polyaromatic hydrocarbons are associated with adult emphysema, chronic bronchitis, asthma, and infections: US NHANES, 2011-2012. Environmental Science and Pollution Research, 23, 25494-25500.
[CrossRef] [Google Scholar] - Guo, J., Wan, Z., Cui, G., Pan, A., & Liu, G. (2021). Association of exposure to ethylene oxide with risk of diabetes mellitus: results from NHANES 2013–2016. Environmental Science and Pollution Research, 28(48), 68551-68559.
[CrossRef] [Google Scholar] - Yin, T., Xu, F., Shi, S., Liao, S., Tang, X., Zhang, H., ... & Li, X. (2022). Vitamin D mediates the association between acrylamide hemoglobin biomarkers and obesity. Environmental Science and Pollution Research, 29(12), 17162-17172.
[CrossRef] [Google Scholar] - Nehme, R., Nahas, P. C., & de Oliveira, E. P. (2023). Association between serum uric acid and muscle strength in older adults with or without gout diagnosis: NHANES 2011-2014. Aging Clinical and Experimental Research, 35(9), 1967-1972.
[CrossRef] [Google Scholar] - Kroenke, K., Spitzer, R. L., & Williams, J. B. W. (2001). The PHQ-9: Validity of a brief depression severity measure. Journal of General Internal Medicine, 16(9), 606-613.
[CrossRef] [Google Scholar] - Spitzer, R. L., Kroenke, K., Williams, J. B., & Patient Health Questionnaire Primary Care Study Group. (1999). Validation and utility of a self-report version of PRIME-MD: the PHQ primary care study. Jama, 282(18), 1737-1744.
[CrossRef] [Google Scholar] - Hou, K., Song, W., He, J., & Ma, Z. (2024). The association between non-high-density lipoprotein cholesterol to high-density lipoprotein cholesterol ratio (NHHR) and prevalence of periodontitis among US adults: a cross-sectional NHANES study. Scientific Reports, 14(1), 5558.
[CrossRef] [Google Scholar] - Lin, L., Xie, Y., Lin, Z., Lin, C., & Yang, Y. (2025). Machine learning for predicting metabolic-associated fatty liver disease including NHHR: a cross-sectional NHANES study. Plos one, 20(3), e0319851.
[CrossRef] [Google Scholar] - Sheng, G., Liu, D., Kuang, M., Zhong, Y., & Zhang, Y. (2022). Utility of non-high-density lipoprotein cholesterol to high-density lipoprotein cholesterol ratio in evaluating incident diabetes risk. Diabetes, Metabolic Syndrome and Obesity: Targets and Therapy, 15, 1677-1686.
[CrossRef] [Google Scholar] - Liu, B., Wang, J., Li, Y. Y., Li, K. P., & Zhang, Q. (2023). The association between systemic immune-inflammation index and rheumatoid arthritis: evidence from NHANES 1999–2018. Arthritis research & therapy, 25(1), 34.
[CrossRef] [Google Scholar] - Dye, B. A., Afful, J., Thornton-Evans, G., & Iafolla, T. (2019). Overview and quality assurance for the oral health component of the National Health and Nutrition Examination Survey (NHANES), 2011–2014. BMC Oral Health, 19(1), 95.
[CrossRef] [Google Scholar] - Le, C. H. H. (2016). The prevalence of anemia and moderate-severe anemia in the US population (NHANES 2003-2012). PloS one, 11(11), e0166635.
[CrossRef] [Google Scholar] - Steele, C. C., Steele, T. J., Rosenkranz, S. K., Lee, J., & Ade, C. J. (2021). Health behaviors and patient–practitioner communication in cancer patients and the general population: an analysis of the National Health and Nutrition Examination Survey (NHANES) 2005–2014. Supportive Care in Cancer, 29(7), 3877-3884.
[CrossRef] [Google Scholar] - Zhu, M., & Chen, A. (2024). Epidemiological characteristics of asthma-COPD overlap, its association with all-cause mortality, and the mediating role of depressive symptoms: Evidence from NHANES 2005-2018. BMC Public Health, 24(1), 1423.
[CrossRef] [Google Scholar] - Xing, W., Gao, W., Zhao, Z., Xu, X., Bu, H., Su, H., ... & Chen, J. (2023). Dietary flavonoids intake contributes to delay biological aging process: analysis from NHANES dataset. Journal of Translational Medicine, 21(1), 492.
[CrossRef] [Google Scholar] - Levine, M. E., Lu, A. T., Quach, A., Chen, B. H., Assimes, T. L., Bandinelli, S., ... & Horvath, S. (2018). An epigenetic biomarker of aging for lifespan and healthspan. Aging (albany NY), 10(4), 573.
[CrossRef] [Google Scholar] - Klemera, P., & Doubal, S. (2006). A new approach to the concept and computation of biological age. Mechanisms of ageing and development, 127(3), 240-248.
[CrossRef] [Google Scholar] - Liu, Z., Kuo, P.-L., Horvath, S., Crimmins, E., Ferrucci, L., & Levine, M. (2018). A new aging measure captures morbidity and mortality risk across diverse subpopulations from NHANES IV: A cohort study. PLoS Medicine, 15(12), e1002718.
[CrossRef] [Google Scholar] - Chen, T. C., Parker, J. D., Clark, J., Shin, H. C., Rammon, J. R., & Burt, V. L. (2018). National Health and Nutrition Examination Survey: Estimation Procedures, 2011-2014. Vital and health statistics. Series 2, Data evaluation and methods research, (177), 1-26.
[Google Scholar] - Chen, T. C., Clark, J., Riddles, M. K., Mohadjer, L. K., & Fakhouri, T. H. I. (2020). National Health and Nutrition Examination Survey, 2015-2018: Sample Design and Estimation Procedures. Vital and Health statistics. Series 2, Data Evaluation and Methods Research, (184), 1-35.
[Google Scholar] - Schober, P., Mascha, E. J., & Vetter, T. R. (2021). Statistics from A (agreement) to Z (z score): A guide to interpreting common measures of association, agreement, diagnostic accuracy, effect size, heterogeneity, and reliability in medical research. Anesthesia & Analgesia, 133(6), 1633-1641.
[CrossRef] [Google Scholar] - Muggeo, V. M., & Muggeo, M. V. M. (2017). Package ‘segmented’. Biometrika, 58(525-534), 516.
[Google Scholar] - Gauthier, J., Wu, Q. V., & Gooley, T. A. (2020). Cubic splines to model relationships between continuous variables and outcomes: A guide for clinicians. Bone Marrow Transplantation, 55(4), 675-680.
[CrossRef] [Google Scholar] - De Winter, J. C. F., Gosling, S. D., & Potter, J. (2016). Comparing the Pearson and Spearman correlation coefficients across distributions and sample sizes: A tutorial using simulations and empirical data. Psychological Methods, 21(3), 273-290.
[CrossRef] [Google Scholar] - Carrico, C., Gennings, C., Wheeler, D. C., & Factor-Litvak, P. (2015). Characterization of weighted quantile sum regression for highly correlated data in a risk analysis setting. Journal of Agricultural, Biological, and Environmental Statistics, 20(1), 100-120.
[CrossRef] [Google Scholar] - LaValley, M. P. (2008). Logistic regression. Circulation, 117(18), 2395-2399.
[CrossRef] [Google Scholar] - Lin, D. Y., & Wei, L. J. (1989). The robust inference for the Cox proportional hazards model. Journal of the American statistical Association, 84(408), 1074-1078.
[CrossRef] [Google Scholar] - Tibshirani, R. (1996). Regression shrinkage and selection via the lasso. Journal of the Royal Statistical Society Series B: Statistical Methodology, 58(1), 267-288.
[CrossRef] [Google Scholar] - Bobb, J. F., Valeri, L., Claus Henn, B., Christiani, D. C., Wright, R. O., Mazumdar, M., ... & Coull, B. A. (2015). Bayesian kernel machine regression for estimating the health effects of multi-pollutant mixtures. Biostatistics, 16(3), 493-508.
[CrossRef] [Google Scholar] - Benoit, L., Koual, M., Tomkiewicz, C., Bats, A. S., Antignac, J. P., Coumoul, X., ... & Cano-Sancho, G. (2022). Impact of mixtures of persistent organic pollutants on breast cancer aggressiveness. Environment International, 170, 107615.
[CrossRef] [Google Scholar] - DeVore, G. R. (2017). Computing the Z score and centiles for cross-sectional analysis: A practical approach. Journal of Ultrasound in Medicine, 36(3), 459-473.
[CrossRef] [Google Scholar] - R Core Team. (2021). R: A language and environment for statistical computing [Computer software]. R Foundation for Statistical Computing.
[Google Scholar] - Chen, W. Y., Fu, Y. P., Tu, H., Zhong, W., & Zhou, L. (2023). The association between exposure to volatile organic compounds and serum lipids in the US adult population. Lipids in Health and Disease, 22(1), 129.
[CrossRef] [Google Scholar] - Prashanth, L., Kattapagari, K. K., Chitturi, R. T., Baddam, V. R. R., & Prasad, L. K. (2015). A review on role of essential trace elements in health and disease. Journal of dr ntr university of health sciences, 4(2), 75-85.
[CrossRef] [Google Scholar] - Kuo, C. L., Pilling, L. C., Liu, Z., Atkins, J. L., & Levine, M. E. (2021). Genetic associations for two biological age measures point to distinct aging phenotypes. Aging Cell, 20(6), e13376.
[CrossRef] [Google Scholar]
Cite This Article
TY - JOUR AU - Meng, Lidian AU - Zheng, He PY - 2026 DA - 2026/03/21 TI - A Data-driven Framework for Modeling Environmental Exposure Mixtures, Biological Aging Acceleration, and Chronic Disease Risk in U.S. Adults JO - Biomedical Informatics and Smart Healthcare T2 - Biomedical Informatics and Smart Healthcare JF - Biomedical Informatics and Smart Healthcare VL - 2 IS - 1 SP - 38 EP - 61 DO - 10.62762/BISH.2026.503823 UR - https://www.icck.org/article/abs/BISH.2026.503823 KW - environmental pollution KW - aging KW - phenotypic age KW - biological age KW - disease risk AB - Environmental exposure biomarkers (EEBs) reflect the internal burden of pollutants, yet the joint effects of multiple exposures on biological aging and chronic disease risk remain insufficiently characterized. We analyzed 8,582 adults from the 2013-2016 National Health and Nutrition Examination Survey (NHANES). Mixed exposure was characterized using 74 EEBs. Phenotypic age acceleration and biological age acceleration were used as aging outcomes. Weighted quantile sum (WQS) regression, Bayesian kernel machine regression (BKMR), and LASSO regression were applied to identify key exposure components associated with aging acceleration. Logistic and Cox regression models were then used to evaluate the associations between aging indicators and chronic disease risks. Higher mixed EEB exposure was significantly associated with accelerated aging, reflected by increases in both phenotypic age and biological age. WQS models identified arsenobetaine, copper, and tin as major contributors to phenotypic age acceleration, whereas selenium, zinc, and MHNCH contributed most strongly to biological age acceleration. Moreover, each one-year increase in phenotypic age was associated with a 30.0\% higher risk of dyslipidemia and a 14.3\% higher risk of metabolic-associated fatty liver disease, while each one-year increase in biological age was associated with a 59.7\% higher risk of chronic obstructive pulmonary disease and a 48.5\% higher risk of anemia. This study proposes a unified data-driven analytical framework that integrates exposure mixtures, biological aging, and disease risk modeling. The findings highlight the importance of evaluating mixed exposures rather than single pollutants and may support risk stratification and prevention strategies in environmental health. SN - 3068-5524 PB - Institute of Central Computation and Knowledge LA - English ER -
@article{Meng2026A,
author = {Lidian Meng and He Zheng},
title = {A Data-driven Framework for Modeling Environmental Exposure Mixtures, Biological Aging Acceleration, and Chronic Disease Risk in U.S. Adults},
journal = {Biomedical Informatics and Smart Healthcare},
year = {2026},
volume = {2},
number = {1},
pages = {38-61},
doi = {10.62762/BISH.2026.503823},
url = {https://www.icck.org/article/abs/BISH.2026.503823},
abstract = {Environmental exposure biomarkers (EEBs) reflect the internal burden of pollutants, yet the joint effects of multiple exposures on biological aging and chronic disease risk remain insufficiently characterized. We analyzed 8,582 adults from the 2013-2016 National Health and Nutrition Examination Survey (NHANES). Mixed exposure was characterized using 74 EEBs. Phenotypic age acceleration and biological age acceleration were used as aging outcomes. Weighted quantile sum (WQS) regression, Bayesian kernel machine regression (BKMR), and LASSO regression were applied to identify key exposure components associated with aging acceleration. Logistic and Cox regression models were then used to evaluate the associations between aging indicators and chronic disease risks. Higher mixed EEB exposure was significantly associated with accelerated aging, reflected by increases in both phenotypic age and biological age. WQS models identified arsenobetaine, copper, and tin as major contributors to phenotypic age acceleration, whereas selenium, zinc, and MHNCH contributed most strongly to biological age acceleration. Moreover, each one-year increase in phenotypic age was associated with a 30.0\\% higher risk of dyslipidemia and a 14.3\\% higher risk of metabolic-associated fatty liver disease, while each one-year increase in biological age was associated with a 59.7\\% higher risk of chronic obstructive pulmonary disease and a 48.5\\% higher risk of anemia. This study proposes a unified data-driven analytical framework that integrates exposure mixtures, biological aging, and disease risk modeling. The findings highlight the importance of evaluating mixed exposures rather than single pollutants and may support risk stratification and prevention strategies in environmental health.},
keywords = {environmental pollution, aging, phenotypic age, biological age, disease risk},
issn = {3068-5524},
publisher = {Institute of Central Computation and Knowledge}
}
Article Metrics
Publisher's Note
ICCK stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and Permissions
 Copyright © 2026 by the Author(s). Published by Institute of Central Computation and Knowledge. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made.
Copyright © 2026 by the Author(s). Published by Institute of Central Computation and Knowledge. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made.

Portico

