Effect of High-voltage Electrostatic Field on Potassium and Phosphate Uptake by Cucumber Plants

Article Information
Abstract
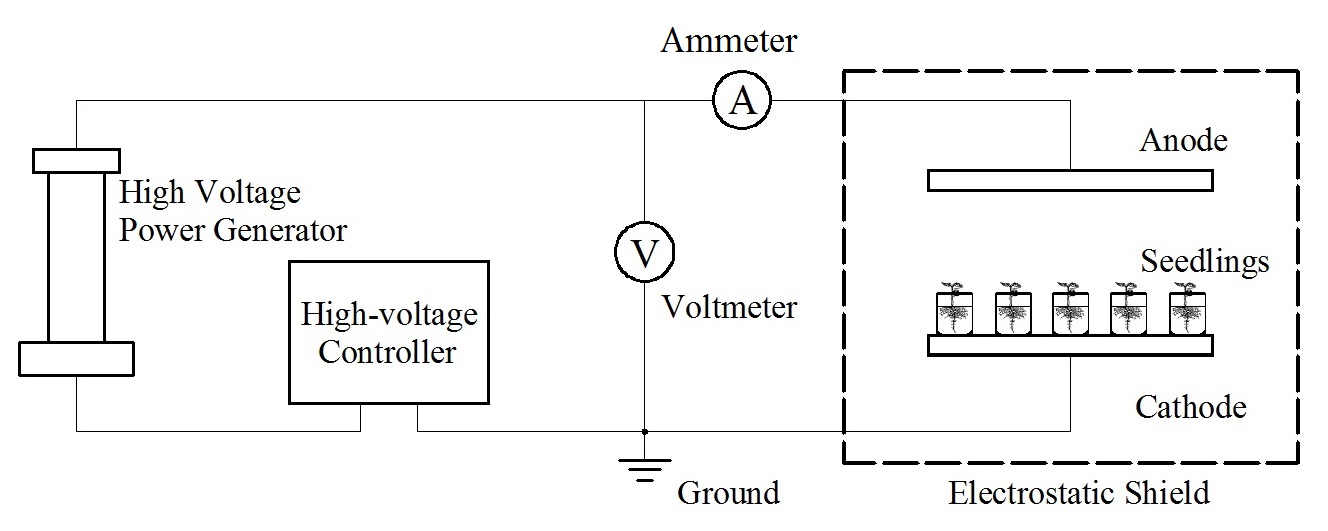
An individual plant cell functions as a complex dynamic circuit, composed of resistor, capacitor, and inductor networks created by cellular structures and macromolecules. Furthermore, high-voltage electrostatic fields (HVEF) alter its electrical properties and initiate metabolic changes. This study aimed to examine the impact of HVEF on the kinetic properties of potassium (K$^+$) and phosphate (H$_2$PO$_4^-$) absorption by Cucumis sativus L. seedlings, and further probed the regulatory mechanisms by constructing a cell membrane potential model. Experimental findings revealed that HVEF substantially altered the maximum uptake rate ($I_{max}$), half-saturation constant ($K_m$), and minimum critical concentration ($C_{min}$) of K$^+$ and H$_2$PO$_4^-$ absorption in cucumber seedlings. Additionally, the regulatory effects displayed a clear dependence on the intensity of the HVEF. Potassium uptake increased as the intensity rose to 1 kV/cm, before declining between 1 and 2 kV/cm. Likewise, phosphate uptake increased as the intensity reached 0.5 kV/cm, before decreasing between 0.5 and 2 kV/cm. A spherical cell model, based on Poisson's equation, indicated that HVEF caused asymmetric membrane potential shifts (hyperpolarization on the lower hemisphere and depolarization on the upper hemisphere), controlling the opening and closing of ion channels. This research offers a theoretical foundation for the use of HVEF in modulating plant nutrient uptake.
Graphical Abstract
Keywords
Data Availability Statement
Funding
Conflicts of Interest
AI Use Statement
Ethical Approval and Consent to Participate
References
- Wu, Y., & Xing, D. (2025). Inaugural editorial for the Journal of Plant Electrobiology. Journal of Plant Electrobiology, 1(1), 1–6.
[CrossRef] [Google Scholar] - Murr, L. E. (1963). Plant growth response in a simulated electric field-environment. Nature, 200(4905), 490-491.
[CrossRef] [Google Scholar] - Nechitailo, G., & Gordeev, A. (2001). Effect of artificial electric fields on plants grown under microgravity conditions. Advances in Space Research, 28(4), 629–631.
[CrossRef] [Google Scholar] - Moon, J. D., & Chung, H. S. (2000). Acceleration of germination of tomato seed by applying AC electric and magnetic fields. Journal of Electrostatics, 48(2), 103–114.
[CrossRef] [Google Scholar] - Sidaway, G. H. (1966). Influence of electrostatic fields on seed germination. Nature, 211(5046), 303-303.
[CrossRef] [Google Scholar] - Sidaway, G. H., & Asprey, G. F. (1968). Influence of electrostatic fields on plant respiration. International Journal of Biometeorology, 12(4), 321-329.
[CrossRef] [Google Scholar] - Li, M. Q., Wu, Y. Y., Zhang, M. M., & Zhu, J. Y. (2018). High-voltage electrostatic fields increase nitrogen uptake and improve growth of tomato seedlings. Canadian Journal of Plant Science, 98(1), 93-106.
[CrossRef] [Google Scholar] - Wu, Y., Zhu, J., Li, Y., & Li, M. (2016). Effect of high-voltage electrostatic field on inorganic nitrogen uptake by cucumber plants. Transactions of the ASABE, 59(1), 25-29.
[CrossRef] [Google Scholar] - Maathuis, F. J., Ichida, A. M., Sanders, D., & Schroeder, J. I. (1997). Roles of higher plant K+ channels. Plant Physiology, 114(4), 1141.
[CrossRef] [Google Scholar] - Schachtman, D. P., Reid, R. J., & Ayling, S. M. (1998). Phosphorus uptake by plants: from soil to cell. Plant physiology, 116(2), 447-453.
[CrossRef] [Google Scholar] - Gross, D., Loew, L. M., & Webb, W. W. (1986). Optical imaging of cell membrane potential changes induced by applied electric fields. Biophysical journal, 50(2), 339-348.
[CrossRef] [Google Scholar] - Zhu, J. K. (2003). Regulation of ion homeostasis under salt stress. Current opinion in plant biology, 6(5), 441-445.
[CrossRef] [Google Scholar] - Etherton, B., & Higinbotham, N. (1960). Transmembrane potential measurements of cells of higher plants as related to salt uptake. Science, 131(3398), 409-410.
[CrossRef] [Google Scholar] - Gassmann, W., & Schroeder, J. I. (1994). Inward-rectifying K+ channels in root hairs of wheat (a mechanism for aluminum-sensitive low-affinity K+ uptake and membrane potential control). Plant Physiology, 105(4), 1399-1408.
[CrossRef] [Google Scholar] - Ullrich, W. R., & Novacky, A. (1981). Nitrate-dependent membrane potential changes and their induction in Lemna gibba G 1. Plant Science Letters, 22(3), 211-217.
[CrossRef] [Google Scholar] - Siddiqi, M. Y., Glass, A. D. M., Ruth, T. J., & Rufty, T. W. (1990). Studies of the uptake of nitrate in barley I. Kinetics of $^{13$NO$_3^-$ influx. Plant Physiology, 93(4), 1426–1432.
[CrossRef] [Google Scholar] - McClure, P. R., Kochian, L. V., Spanswick, R. M., & Shaff, J. E. (1990). Evidence for cotransport of nitrate and protons in maize roots: I. Effects of nitrate on the membrane potential. Plant Physiology, 93(1), 281-289.
[CrossRef] [Google Scholar] - McClure, P. R., Kochian, L. V., Spanswick, R. M., & Shaff, J. E. (1990). Evidence for cotransport of nitrate and protons in maize roots II. Measurement of NO$_3^-$ and H+ fluxes with ion-selective microelectrodes. Plant Physiology, 93(1), 290–294.
[CrossRef] [Google Scholar] - Iqbal, B., Zhou, H., Raheem, A., Inamullah, Zheng, T., Khan, A. R., ... & Du, D. (2025). The Impact of Land Use Changes and Agricultural Practices on Phosphorus Cycling: A Review. Land Degradation & Development.
[CrossRef] [Google Scholar] - Iqbal, B., Khan, I., Javed, Q., Alabbosh, K. F., Inamullah, I., Zhou, Z., & Rehman, A. (2023). The high phosphorus incorporation promotes the soil enzymatic activity, nutritional status, and biomass of the crop. Polish Journal of Environmental Studies, 32(3), 2125-2139.
[CrossRef] [Google Scholar] - Fu, S., Iqbal, B., Alahmadi, T. A., Ansari, M. J., Jalal, A., Nazir, M. J., Li, G., & Du, D. (2025). Ammonium nitrate enhances salinity stress tolerance by inducing physiological and phytohormonal regulation changes in Vigna radiata L. Global Nest Journal, 27(6), 07514.
[Google Scholar] - Khan, A. A., Alabbosh, K. F., Kashif, Iqbal, B., Manan, S., Alhoqail, W. A., ... & Wang, Y. F. (2025). Ion Homeostasis, Osmotic Adjustment, and ROS Detoxification Underlie Pea Salinity Tolerance Induced by Pseudomonas putida RT12. Microbiology Research, 16(11), 227.
[CrossRef] [Google Scholar] - Drew, M. C., Saker, L. R., Barber, S. A., & Jenkins, W. (1984). Changes in the kinetics of phosphate and potassium absorption in nutrient-deficient barley roots measured by a solution-depletion technique. Planta, 160(6), 490–499.
[CrossRef] [Google Scholar] - Down, J. E., & Riggs, D. S. (1965). A comparison of estimates of Michaelis-Menten kinetic constants from various linear transformations. Journal of Biological Chemistry, 240(2), 863–869.
[Google Scholar] - Cole, K. S. (1972). Membranes, ions and impulses: a chapter of classical biophysics (Vol. 1). Univ of California Press.
[Google Scholar] - Hille, B. (1978). Ionic channels in excitable membranes. Current problems and biophysical approaches. Biophysical journal, 22(2), 283-294.
[CrossRef] [Google Scholar] - Doyle, D. A., Cabral, J. M., Pfuetzner, R. A., Kuo, A., Gulbis, J. M., Cohen, S. L., ... & MacKinnon, R. (1998). The structure of the potassium channel: molecular basis of K+ conduction and selectivity. science, 280(5360), 69-77.
[CrossRef] [Google Scholar] - Jaffe, L. F. (1977). Electrophoresis along cell membranes. Nature, 265(5595), 600-602.
[CrossRef] [Google Scholar] - Poo, M. M. (1981). In situ electrophoresis of membrane components. Annual Review of Biophysics and Bioengineering, 10, 245–276.
[CrossRef] [Google Scholar] - Gross, D. A. V. I. D. (1988). Electromobile surface charge alters membrane potential changes induced by applied electric fields. Biophysical journal, 54(5), 879-884.
[CrossRef] [Google Scholar] - Teissie, J., & Rols, M. P. (1993). An experimental evaluation of the critical potential difference inducing cell membrane electropermeabilization. Biophysical journal, 65(1), 409-413.
[CrossRef] [Google Scholar] - Lojewska, Z., Farkas, D. L., Ehrenberg, B., & Loew, L. M. (1989). Analysis of the effect of medium and membrane conductance on the amplitude and kinetics of membrane potentials induced by externally applied electric fields. Biophysical journal, 56(1), 121-128.
[CrossRef] [Google Scholar] - Tekle, E., Astumian, R. D., & Chock, P. B. (1990). Electro-permeabilization of cell membranes: effect of the resting membrane potential. Biochemical and biophysical research communications, 172(1), 282-287.
[CrossRef] [Google Scholar] - Hibino, M., Shigemori, M., Itoh, H., Nagayama, K., & Kinosita, K. (1991). Membrane conductance of an electroporated cell analyzed by submicrosecond imaging of transmembrane potential. Biophysical journal, 59(1), 209-220.
[CrossRef] [Google Scholar] - Teissié, J., Eynard, N., Gabriel, B., & Rols, M. P. (1999). Electropermeabilization of cell membranes. Advanced drug delivery reviews, 35(1), 3-19.
[CrossRef] [Google Scholar] - Neumann, E., Sowers, A. E., & Jordan, C. A. (1989). Electroporation and electrofusion in cell biology (pp. 59–164). Springer.
[CrossRef] [Google Scholar] - Zimmermann, U., & Neil, G. A. (1996). Electromanipulation of cells (pp. 1–106). CRC Press.
[Google Scholar] - Weaver, J. C. (2002). Electroporation of cells and tissues. IEEE transactions on plasma science, 28(1), 24-33.
[CrossRef] [Google Scholar] - Schoenbach, K. H., Joshi, R. P., Kolb, J. F., Chen, N., Stacey, M., Blackmore, P. F., ... & Beebe, S. J. (2004). Ultrashort electrical pulses open a new gateway into biological cells. Proceedings of the IEEE, 92(7), 1122-1137.
[CrossRef] [Google Scholar] - Chapel, M., Teissié, J., & Alibert, G. (1984). Electrofusion of spermine-treated plant protoplasts. FEBS Letters, 173(2), 331–336.
[CrossRef] [Google Scholar] - Puite, K. J. (1992). Progress in plant protoplast research. Physiologia Plantarum, 85(2), 403–410.
[CrossRef] [Google Scholar] - Glaser, R. W., Leikin, S. L., Chernomordik, L. V., Pastushenko, V. F., & Sokirko, A. I. (1988). Reversible electrical breakdown of lipid bilayers: formation and evolution of pores. Biochimica et Biophysica Acta (BBA)-Biomembranes, 940(2), 275-287.
[CrossRef] [Google Scholar] - Gowrishankar, T. R., Chen, W., & Lee, R. C. (1998). Non‐Linear Microscale Alterations in Membrane Transport by Electropermeabilization a. Annals of the New York Academy of Sciences, 858(1), 205-216.
[CrossRef] [Google Scholar] - Maathuis, F. J., & Sanders, D. (1996). Mechanisms of potassium absorption by higher plant roots. Physiologia Plantarum, 96(1), 158-168.
[CrossRef] [Google Scholar]
Cite This Article
TY - JOUR
AU - Zhu, Jianyun
AU - Li, Meiqing
PY - 2026
DA - 2026/03/11
TI - Effect of High-voltage Electrostatic Field on Potassium and Phosphate Uptake by Cucumber Plants
JO - Journal of Plant Electrobiology
T2 - Journal of Plant Electrobiology
JF - Journal of Plant Electrobiology
VL - 1
IS - 1
SP - 32
EP - 41
DO - 10.62762/JPE.2025.597638
UR - https://www.icck.org/article/abs/JPE.2025.597638
KW - Cucumis sativus L.
KW - depolarization
KW - high-voltage electric field
KW - hyperpolarization
KW - ion absorption kinetics
AB - An individual plant cell functions as a complex dynamic circuit, composed of resistor, capacitor, and inductor networks created by cellular structures and macromolecules. Furthermore, high-voltage electrostatic fields (HVEF) alter its electrical properties and initiate metabolic changes. This study aimed to examine the impact of HVEF on the kinetic properties of potassium (K$^+$) and phosphate (H$_2$PO$_4^-$) absorption by Cucumis sativus L. seedlings, and further probed the regulatory mechanisms by constructing a cell membrane potential model. Experimental findings revealed that HVEF substantially altered the maximum uptake rate ($I_{max}$), half-saturation constant ($K_m$), and minimum critical concentration ($C_{min}$) of K$^+$ and H$_2$PO$_4^-$ absorption in cucumber seedlings. Additionally, the regulatory effects displayed a clear dependence on the intensity of the HVEF. Potassium uptake increased as the intensity rose to 1 kV/cm, before declining between 1 and 2 kV/cm. Likewise, phosphate uptake increased as the intensity reached 0.5 kV/cm, before decreasing between 0.5 and 2 kV/cm. A spherical cell model, based on Poisson's equation, indicated that HVEF caused asymmetric membrane potential shifts (hyperpolarization on the lower hemisphere and depolarization on the upper hemisphere), controlling the opening and closing of ion channels. This research offers a theoretical foundation for the use of HVEF in modulating plant nutrient uptake.
SN - pending
PB - Institute of Central Computation and Knowledge
LA - English
ER -
@article{Zhu2026Effect,
author = {Jianyun Zhu and Meiqing Li},
title = {Effect of High-voltage Electrostatic Field on Potassium and Phosphate Uptake by Cucumber Plants},
journal = {Journal of Plant Electrobiology},
year = {2026},
volume = {1},
number = {1},
pages = {32-41},
doi = {10.62762/JPE.2025.597638},
url = {https://www.icck.org/article/abs/JPE.2025.597638},
abstract = {An individual plant cell functions as a complex dynamic circuit, composed of resistor, capacitor, and inductor networks created by cellular structures and macromolecules. Furthermore, high-voltage electrostatic fields (HVEF) alter its electrical properties and initiate metabolic changes. This study aimed to examine the impact of HVEF on the kinetic properties of potassium (K\$^+\$) and phosphate (H\$\_2\$PO\$\_4^-\$) absorption by Cucumis sativus L. seedlings, and further probed the regulatory mechanisms by constructing a cell membrane potential model. Experimental findings revealed that HVEF substantially altered the maximum uptake rate (\$I\_{max}\$), half-saturation constant (\$K\_m\$), and minimum critical concentration (\$C\_{min}\$) of K\$^+\$ and H\$\_2\$PO\$\_4^-\$ absorption in cucumber seedlings. Additionally, the regulatory effects displayed a clear dependence on the intensity of the HVEF. Potassium uptake increased as the intensity rose to 1 kV/cm, before declining between 1 and 2 kV/cm. Likewise, phosphate uptake increased as the intensity reached 0.5 kV/cm, before decreasing between 0.5 and 2 kV/cm. A spherical cell model, based on Poisson's equation, indicated that HVEF caused asymmetric membrane potential shifts (hyperpolarization on the lower hemisphere and depolarization on the upper hemisphere), controlling the opening and closing of ion channels. This research offers a theoretical foundation for the use of HVEF in modulating plant nutrient uptake.},
keywords = {Cucumis sativus L., depolarization, high-voltage electric field, hyperpolarization, ion absorption kinetics},
issn = {pending},
publisher = {Institute of Central Computation and Knowledge}
}
Article Metrics
Publisher's Note
ICCK stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


