Oncology Communications
ISSN: pending (Online)
Email: [email protected]
 Submit Manuscript
Edit a Special Issue
Submit Manuscript
Edit a Special Issue
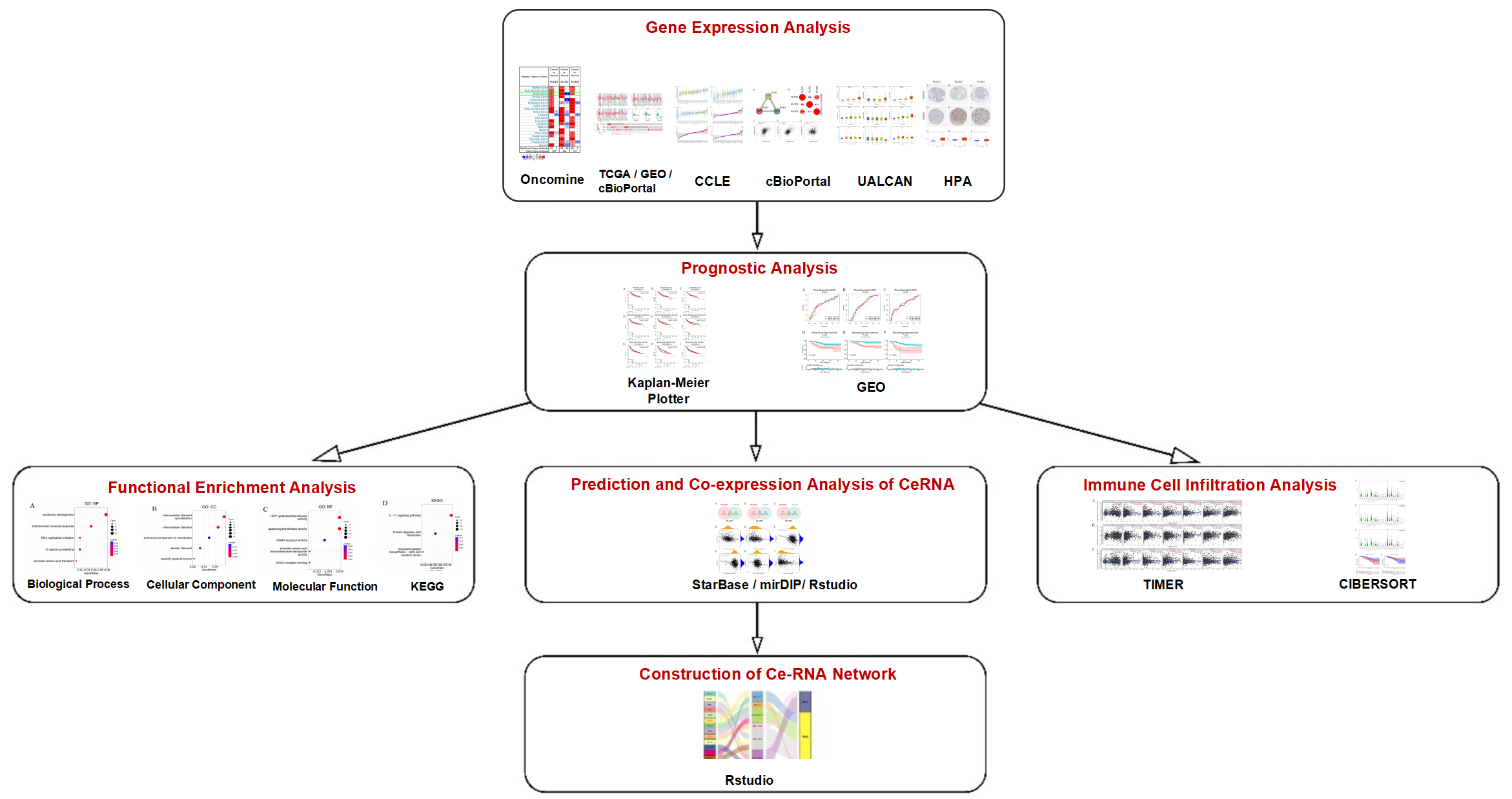
TY - JOUR AU - Li, Jinyang AU - Zhou, Chuqiao AU - Anwar, Munawar AU - Xia, Linxiao AU - Qu, Limeng PY - 2026 DA - 2026/02/27 TI - Comprehensive Analysis of PLOD Family Prognostic Value and Related Regulatory ceRNA Network in Breast Cancer JO - Oncology Communications T2 - Oncology Communications JF - Oncology Communications VL - 1 IS - 1 SP - 20 EP - 42 DO - 10.62762/OC.2025.804127 UR - https://www.icck.org/article/abs/OC.2025.804127 KW - PLOD family KW - ceRNA network KW - breast cancer KW - prognostic AB - Procollagen-lysine, 2-oxoglutarate 5-dioxygenases (PLODs) catalyze lysine hydroxylation, promoting collagen crosslinking and extracellular matrix stability, and are implicated in tumor aggressiveness. However, their expression and prognostic value in breast cancer (BC) remain unclear. We explored PLOD1-3 expression in BC using ONCOMINE, TIMER, CCLE, cBioPortal, UALCAN, GEPIA, and HPA. Prognostic associations were assessed via Kaplan-Meier Plotter, with enrichment analysis by clusterProfiler. A competing endogenous RNA (ceRNA) network was constructed using TCGA data, and immune infiltration analyzed by TIMER and CIBERSORT. PLOD1-3 were upregulated in BC versus normal tissues at both transcript and protein levels, with high expression predicting shorter survival. Enrichment analysis implicated PLODs in DNA replication, cytokinesis, and basement membrane formation. The ceRNA network for PLODs was successfully constructed. Immune infiltration analysis revealed significant correlations between PLOD expression and immune cell levels; high M2 macrophage or low plasma cell infiltration indicated poor prognosis. PLODs are highly expressed in BC and may serve as prognostic biomarkers and therapeutic targets. SN - pending PB - Institute of Central Computation and Knowledge LA - English ER -
@article{Li2026Comprehens,
author = {Jinyang Li and Chuqiao Zhou and Munawar Anwar and Linxiao Xia and Limeng Qu},
title = {Comprehensive Analysis of PLOD Family Prognostic Value and Related Regulatory ceRNA Network in Breast Cancer},
journal = {Oncology Communications},
year = {2026},
volume = {1},
number = {1},
pages = {20-42},
doi = {10.62762/OC.2025.804127},
url = {https://www.icck.org/article/abs/OC.2025.804127},
abstract = {Procollagen-lysine, 2-oxoglutarate 5-dioxygenases (PLODs) catalyze lysine hydroxylation, promoting collagen crosslinking and extracellular matrix stability, and are implicated in tumor aggressiveness. However, their expression and prognostic value in breast cancer (BC) remain unclear. We explored PLOD1-3 expression in BC using ONCOMINE, TIMER, CCLE, cBioPortal, UALCAN, GEPIA, and HPA. Prognostic associations were assessed via Kaplan-Meier Plotter, with enrichment analysis by clusterProfiler. A competing endogenous RNA (ceRNA) network was constructed using TCGA data, and immune infiltration analyzed by TIMER and CIBERSORT. PLOD1-3 were upregulated in BC versus normal tissues at both transcript and protein levels, with high expression predicting shorter survival. Enrichment analysis implicated PLODs in DNA replication, cytokinesis, and basement membrane formation. The ceRNA network for PLODs was successfully constructed. Immune infiltration analysis revealed significant correlations between PLOD expression and immune cell levels; high M2 macrophage or low plasma cell infiltration indicated poor prognosis. PLODs are highly expressed in BC and may serve as prognostic biomarkers and therapeutic targets.},
keywords = {PLOD family, ceRNA network, breast cancer, prognostic},
issn = {pending},
publisher = {Institute of Central Computation and Knowledge}
}
 Copyright © 2026 by the Author(s). Published by Institute of Central Computation and Knowledge. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made.
Copyright © 2026 by the Author(s). Published by Institute of Central Computation and Knowledge. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. 
Portico
All published articles are preserved here permanently:
https://www.portico.org/publishers/icck/