ICCK Transactions on Applied Intelligence and Cybernetics
ISSN: pending (Online) | ISSN: pending (Print)
Email: [email protected]
 Submit Manuscript
Edit a Special Issue
Submit Manuscript
Edit a Special Issue
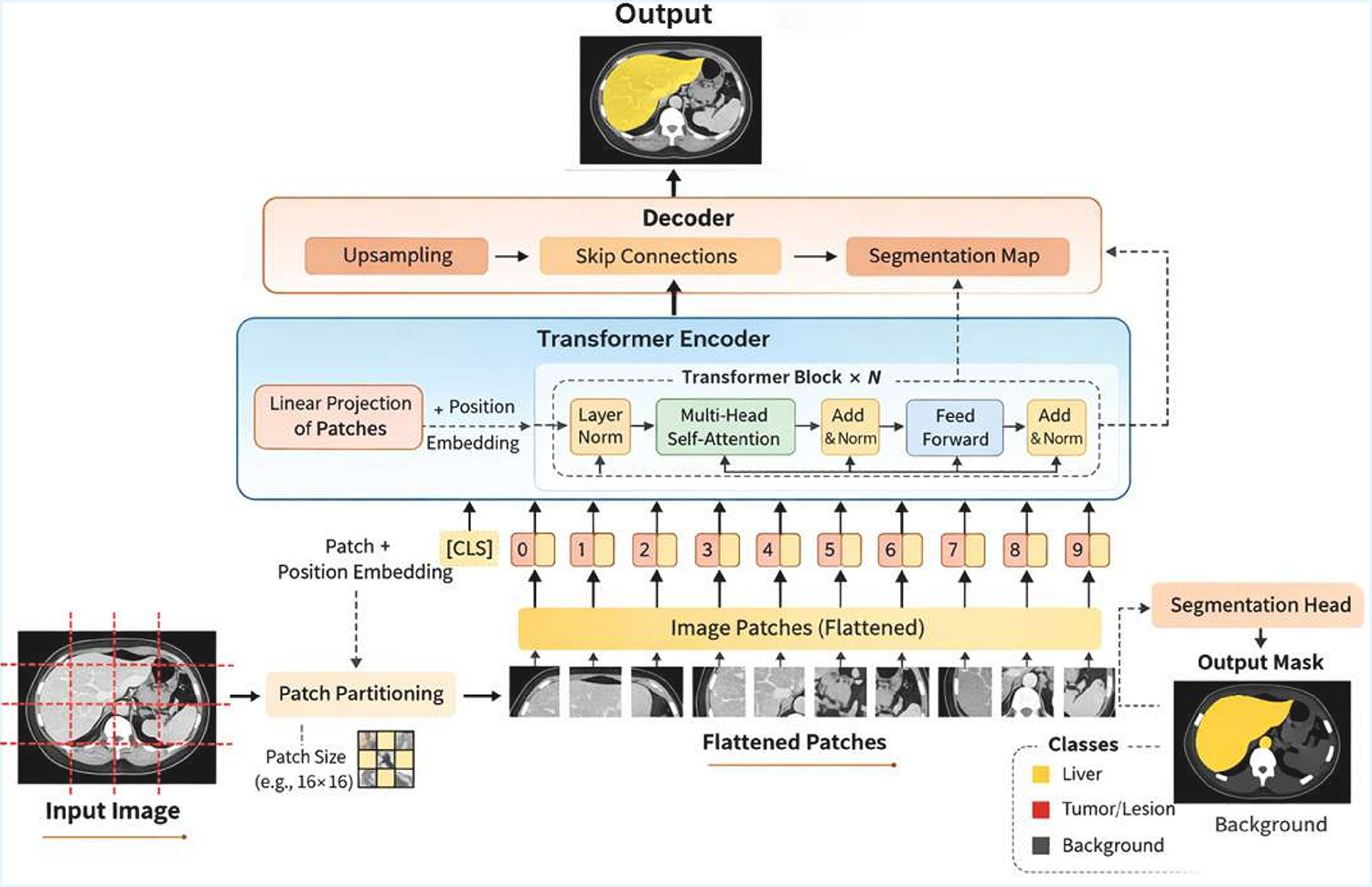
TY - JOUR AU - Khan, Irshad Ali AU - Khan, Gul Zaman AU - Muhammad, Yar AU - Ihsan, Samreen AU - Haq, Ijazul PY - 2026 DA - 2026/03/03 TI - A Comprehensive Review on 3D Volumetric CT Liver Segmentation: Techniques, Challenges, Trends, and Future Research Directions JO - ICCK Transactions on Applied Intelligence and Cybernetics T2 - ICCK Transactions on Applied Intelligence and Cybernetics JF - ICCK Transactions on Applied Intelligence and Cybernetics VL - 1 IS - 1 SP - 5 EP - 35 DO - 10.62762/TAIC.2025.965486 UR - https://www.icck.org/article/abs/TAIC.2025.965486 KW - 3D liver segmentation KW - computed tomography KW - deep learning KW - transformers KW - medical image analysis KW - self-supervised learning AB - Accurate liver segmentation from three-dimensional (3D) computed tomography (CT) volumes is a critical step in computer-aided diagnosis, surgical planning, and disease quantification. Despite substantial progress in deep learning, achieving robust and generalizable liver segmentation remains challenging due to complex organ boundaries, pathological variations, and domain shifts across scanners. This review provides a comprehensive overview of 3D volumetric liver segmentation techniques, spanning from classical model-based methods to contemporary transformer-driven frameworks. We categorize existing methods into three paradigms: (1) classical statistical and atlas-based methods, (2) deep convolutional architectures, and (3) hybrid and attention-based transformer approaches. Key benchmark datasets, evaluation metrics, and performance comparisons are discussed in detail. Furthermore, we highlight open challenges, such as data imbalance, domain generalization, and clinical interpretability, and propose potential future directions, including self-supervised learning, multi-modal integration, and foundation models. Additionally, we identify evolving trends toward dual-stream CNN–Transformer integration, attention-enhanced spatial reasoning, and foundation-model-driven segmentation pipelines. This review aims to serve as a reference for researchers and practitioners seeking to develop next-generation 3D liver segmentation systems. SN - pending PB - Institute of Central Computation and Knowledge LA - English ER -
@article{Khan2026A,
author = {Irshad Ali Khan and Gul Zaman Khan and Yar Muhammad and Samreen Ihsan and Ijazul Haq},
title = {A Comprehensive Review on 3D Volumetric CT Liver Segmentation: Techniques, Challenges, Trends, and Future Research Directions},
journal = {ICCK Transactions on Applied Intelligence and Cybernetics},
year = {2026},
volume = {1},
number = {1},
pages = {5-35},
doi = {10.62762/TAIC.2025.965486},
url = {https://www.icck.org/article/abs/TAIC.2025.965486},
abstract = {Accurate liver segmentation from three-dimensional (3D) computed tomography (CT) volumes is a critical step in computer-aided diagnosis, surgical planning, and disease quantification. Despite substantial progress in deep learning, achieving robust and generalizable liver segmentation remains challenging due to complex organ boundaries, pathological variations, and domain shifts across scanners. This review provides a comprehensive overview of 3D volumetric liver segmentation techniques, spanning from classical model-based methods to contemporary transformer-driven frameworks. We categorize existing methods into three paradigms: (1) classical statistical and atlas-based methods, (2) deep convolutional architectures, and (3) hybrid and attention-based transformer approaches. Key benchmark datasets, evaluation metrics, and performance comparisons are discussed in detail. Furthermore, we highlight open challenges, such as data imbalance, domain generalization, and clinical interpretability, and propose potential future directions, including self-supervised learning, multi-modal integration, and foundation models. Additionally, we identify evolving trends toward dual-stream CNN–Transformer integration, attention-enhanced spatial reasoning, and foundation-model-driven segmentation pipelines. This review aims to serve as a reference for researchers and practitioners seeking to develop next-generation 3D liver segmentation systems.},
keywords = {3D liver segmentation, computed tomography, deep learning, transformers, medical image analysis, self-supervised learning},
issn = {pending},
publisher = {Institute of Central Computation and Knowledge}
}

ICCK Transactions on Applied Intelligence and Cybernetics
ISSN: pending (Online) | ISSN: pending (Print)
Email: [email protected]

Portico
All published articles are preserved here permanently:
https://www.portico.org/publishers/icck/