Bridging Predictive Modeling and Clinical Interpretability: An Explainable AI Approach to Parkinson’s Disease Detection

Article Information
Abstract
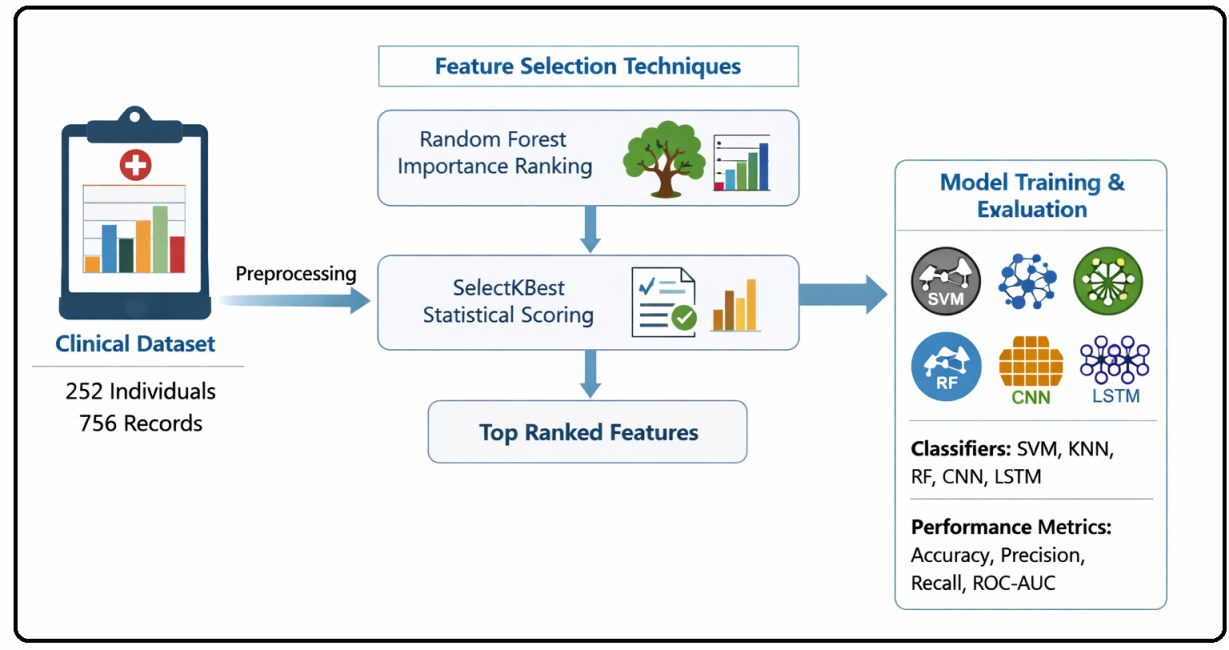
Parkinson’s disease (PD) is the second most common neurodegenerative disorder worldwide, predominantly affecting older adults. Early detection is crucial, as subtle motor and non-motor symptoms frequently overlap with other conditions, often resulting in delayed diagnosis. Many existing models rely on costly and less accessible imaging modalities such as MRI or PET scans, limiting their applicability in resource-constrained settings where only routine clinical data are available. This study develops interpretable AI models for early PD detection using structured clinical variables, incorporating feature selection techniques. Feature selection was conducted via Random Forest (RF) importance ranking combined with SelectKBest statistical scoring, retaining the most informative predictors for modeling. Five classifiers were implemented in parallel: Support Vector Machine (SVM), K-Nearest Neighbors (KNN), RF, Convolutional Neural Network (CNN), and Long Short-Term Memory (LSTM) network. Model performance was assessed using accuracy, precision, recall, F1-score, and ROC-AUC metrics. The RF model achieved the highest accuracy of 92.72% (ROC-AUC: 0.968), with CNN and SVM showing competitive performance. LSTM exhibited balanced sensitivity and specificity, while KNN demonstrated relatively lower recall. To improve clinical interpretability, LIME was applied to each model to produce instance-level explanations, consistently highlighting tremor severity, motor impairment, cognitive scores, and age as key influential features. These results demonstrate that structured clinical variables alone can enable reliable PD detection without dependence on imaging. Integrating explainable artificial intelligence enhances transparency and supports responsible clinical adoption.
Graphical Abstract
Keywords
Data Availability Statement
Funding
Conflicts of Interest
AI Use Statement
Ethical Approval and Consent to Participate
References
- Velu, K., & Jaisankar, N. (2025). Design of an Early Prediction Model for Parkinson's Disease Using Machine Learning. IEEE Access, 13, 17457-17472.
[CrossRef] [Google Scholar] - Dentamaro, V., Impedovo, D., Musti, L., Pirlo, G., & Taurisano, P. (2024). Enhancing early Parkinson's disease detection through multimodal deep learning and explainable AI: insights from the PPMI database. Scientific Reports, 14(1), 20941.
[CrossRef] [Google Scholar] - Priyadharshini, S., Ramkumar, K., Vairavasundaram, S., Narasimhan, K., Venkatesh, S., Amirtharajan, R., & Kotecha, K. (2024). A comprehensive framework for parkinson’s disease diagnosis using explainable artificial intelligence empowered machine learning techniques. Alexandria Engineering Journal, 107, 568-582.
[CrossRef] [Google Scholar] - Tolosa, E., Garrido, A., Scholz, S. W., & Poewe, W. (2021). Challenges in the diagnosis of Parkinson's disease. The Lancet Neurology, 20(5), 385-397.
[CrossRef] [Google Scholar] - Esteva, A., Robicquet, A., Ramsundar, B., Kuleshov, V., DePristo, M., Chou, K., ... & Dean, J. (2019). A guide to deep learning in healthcare. Nature medicine, 25(1), 24-29.
[CrossRef] [Google Scholar] - Sharma, R., Sharma, K. D., & Bijalwan, A. (2025). HDSF: A Healthcare Decision Support Framework to Provide A Seamless and Adaptable Patient Experience. Biomedical Informatics and Smart Healthcare, 1(1), 1-8.
[CrossRef] [Google Scholar] - Sivaranjini, S., & Sujatha, C. M. (2020). Deep learning based diagnosis of Parkinson’s disease using convolutional neural network. Multimedia tools and applications, 79(21), 15467-15479.
[CrossRef] [Google Scholar] - Tewari, Y., Parihar, N. S., Rautela, K., Kaundal, N., Diwakar, M., & Pandey, N. K. (2025). Diabetic retinopathy detection and analysis with convolutional neural networks and vision transformer. Biomedical Informatics and Smart Healthcare, 1(1), 18-26.
[CrossRef] [Google Scholar] - Srinivasan, S., Ramadass, P., Mathivanan, S. K., Panneer Selvam, K., Shivahare, B. D., & Shah, M. A. (2024). Detection of Parkinson disease using multiclass machine learning approach. Scientific Reports, 14(1), 13813.
[CrossRef] [Google Scholar] - Bauskar, S. (2021). An Analysis: Early Diagnosis and Classification of Parkinson's Disease Using Machine Learning Techniques. International Journal of Computer Engineering and Technology, 12(1), 54-66.
[CrossRef] [Google Scholar] - Farooq, W., Ali, A., Fatima, H. M., Rafiq, W., Zainab, N. E., & Ali, M. (2026). FusedCNN-LSTM: A Software-Oriented Multimodal Deep Learning Framework for Intelligent Hypertension Risk Prediction. ICCK Journal of Software Engineering, 2(1), 11-29.
[CrossRef] [Google Scholar] - Wiratsin, I.-O., & Ragkhitwetsagul, C. (2025). Effectiveness of Explainable Artificial Intelligence (XAI) Techniques for Improving Human Trust in Machine Learning Models: A Systematic Literature Review. IEEE Access, 13, 121326-121350.
[CrossRef] [Google Scholar] - Henninger, M., & Strobl, C. (2025). Interpreting machine learning predictions with LIME and Shapley values: theoretical insights, challenges, and meaningful interpretations. Behaviormetrika, 52(1), 45-75.
[CrossRef] [Google Scholar] - Adadi, A., & Berrada, M. (2020). Explainable AI for Healthcare: From Black Box to Interpretable Models. In Proceedings of the International Conference on Emerging Trends in Electrical, Electronic and Communications Engineering (pp. 327-337). Springer.
[CrossRef] [Google Scholar] - Arrieta, A. B., Díaz-Rodríguez, N., Del Ser, J., Bennetot, A., Tabik, S., Barbado, A., ... & Herrera, F. (2020). Explainable Artificial Intelligence (XAI): Concepts, taxonomies, opportunities and challenges toward responsible AI. Information fusion, 58, 82-115.
[CrossRef] [Google Scholar] - Hossain, M. A., Traini, E., & Amenta, F. (2025). Machine learning applications for diagnosing parkinson’s disease via speech, language, and voice changes: A systematic review. Inventions, 10(4), 48.
[CrossRef] [Google Scholar] - Shokrpour, S., MoghadamFarid, A., Bazzaz Abkenar, S., Haghi Kashani, M., Akbari, M., & Sarvizadeh, M. (2025). Machine learning for Parkinson’s disease: a comprehensive review of datasets, algorithms, and challenges. npj Parkinson's Disease, 11(1), 187.
[CrossRef] [Google Scholar] - Nayan, N. M., Rana, A. M., Islam, M. M., Uddin, J., Yasmin, T., & Uddin, J. (2025). An interpretable and balanced machine learning framework for Parkinson's disease prediction using feature engineering and explainable AI. PLOS One, 20(10), e0333418.
[CrossRef] [Google Scholar] - Quan, C., Ren, K., & Luo, Z. (2021). A Deep Learning Based Method for Parkinson's Disease Detection Using Dynamic Features of Speech. IEEE Access, 9, 10239-10252.
[CrossRef] [Google Scholar] - Trabassi, D., Serrao, M., Varrecchia, T., Ranavolo, A., Coppola, G., De Icco, R., ... & Castiglia, S. F. (2022). Machine learning approach to support the detection of Parkinson’s disease in IMU-based gait analysis. Sensors, 22(10), 3700.
[CrossRef] [Google Scholar] - Escobar-Grisales, D., Ríos-Urrego, C. D., & Orozco-Arroyave, J. R. (2023). Deep learning and artificial intelligence applied to model speech and language in Parkinson’s disease. Diagnostics, 13(13), 2163.
[CrossRef] [Google Scholar] - Um, T. T., Pfister, F. M., Pichler, D., Endo, S., Lang, M., Hirche, S., ... & Kulić, D. (2017, November). Data augmentation of wearable sensor data for parkinson’s disease monitoring using convolutional neural networks. In Proceedings of the 19th ACM international conference on multimodal interaction (pp. 216-220).
[CrossRef] [Google Scholar] - Drotár, P., Mekyska, J., Rektorová, I., Masarová, L., Smékal, Z., & Faundez-Zanuy, M. (2016). Evaluation of handwriting kinematics and pressure for differential diagnosis of Parkinson's disease. Artificial Intelligence in Medicine, 67, 39-46.
[CrossRef] [Google Scholar] - Gunduz, H. (2019). Deep Learning-Based Parkinson's Disease Classification Using Vocal Feature Sets. IEEE Access, 7, 115540-115551.
[CrossRef] [Google Scholar] - Yu, J., Meng, K., Liang, T., Liu, H., & Wang, X. (2024). Improved deep learning for Parkinson's diagnosis based on wearable sensors. Electronics, 13(23), 4638.
[CrossRef] [Google Scholar] - Shi, D., Zhang, H., Wang, G., Wang, S., Yao, X., Li, Y., ... & Ren, K. (2022). Machine learning for detecting parkinson’s disease by resting-state functional magnetic resonance imaging: A multicenter radiomics analysis. Frontiers in aging neuroscience, 14, 806828.
[CrossRef] [Google Scholar] - Martinez-Eguiluz, M., Arbelaitz, O., Gurrutxaga, I., Muguerza, J., Perona, I., Murueta-Goyena, A., ... & Gabilondo, I. (2023). Diagnostic classification of Parkinson’s disease based on non-motor manifestations and machine learning strategies. Neural Computing and Applications, 35(8), 5603-5617.
[CrossRef] [Google Scholar] - Moon, S., Song, H. J., Sharma, V. D., Lyons, K. E., Pahwa, R., Akinwuntan, A. E., & Devos, H. (2020). Classification of Parkinson’s disease and essential tremor based on balance and gait characteristics from wearable motion sensors via machine learning techniques: a data-driven approach. Journal of neuroengineering and rehabilitation, 17(1), 125.
[CrossRef] [Google Scholar] - Fu, J., Chen, H., Xu, C., Jia, Z., Lu, Q., Zhang, H., ... & Geng, D. (2025). Harnessing routine MRI for the early screening of Parkinson's disease: a multicenter machine learning study using T2-weighted FLAIR imaging. Insights into Imaging, 16(1), 92-92.
[CrossRef] [Google Scholar]
Cite This Article
TY - JOUR AU - Raza, Aamir AU - Ali, Aamir AU - Kumar, Aashesh AU - Fatima, Nikhat AU - Ali, Misbah PY - 2026 DA - 2026/03/12 TI - Bridging Predictive Modeling and Clinical Interpretability: An Explainable AI Approach to Parkinson’s Disease Detection JO - Biomedical Informatics and Smart Healthcare T2 - Biomedical Informatics and Smart Healthcare JF - Biomedical Informatics and Smart Healthcare VL - 2 IS - 1 SP - 20 EP - 37 DO - 10.62762/BISH.2026.470997 UR - https://www.icck.org/article/abs/BISH.2026.470997 KW - Parkinson’s disease KW - explainable artificial intelligence KW - machine learning KW - deep learning KW - feature selection KW - clinical decision support AB - Parkinson’s disease (PD) is the second most common neurodegenerative disorder worldwide, predominantly affecting older adults. Early detection is crucial, as subtle motor and non-motor symptoms frequently overlap with other conditions, often resulting in delayed diagnosis. Many existing models rely on costly and less accessible imaging modalities such as MRI or PET scans, limiting their applicability in resource-constrained settings where only routine clinical data are available. This study develops interpretable AI models for early PD detection using structured clinical variables, incorporating feature selection techniques. Feature selection was conducted via Random Forest (RF) importance ranking combined with SelectKBest statistical scoring, retaining the most informative predictors for modeling. Five classifiers were implemented in parallel: Support Vector Machine (SVM), K-Nearest Neighbors (KNN), RF, Convolutional Neural Network (CNN), and Long Short-Term Memory (LSTM) network. Model performance was assessed using accuracy, precision, recall, F1-score, and ROC-AUC metrics. The RF model achieved the highest accuracy of 92.72% (ROC-AUC: 0.968), with CNN and SVM showing competitive performance. LSTM exhibited balanced sensitivity and specificity, while KNN demonstrated relatively lower recall. To improve clinical interpretability, LIME was applied to each model to produce instance-level explanations, consistently highlighting tremor severity, motor impairment, cognitive scores, and age as key influential features. These results demonstrate that structured clinical variables alone can enable reliable PD detection without dependence on imaging. Integrating explainable artificial intelligence enhances transparency and supports responsible clinical adoption. SN - 3068-5524 PB - Institute of Central Computation and Knowledge LA - English ER -
@article{Raza2026Bridging,
author = {Aamir Raza and Aamir Ali and Aashesh Kumar and Nikhat Fatima and Misbah Ali},
title = {Bridging Predictive Modeling and Clinical Interpretability: An Explainable AI Approach to Parkinson’s Disease Detection},
journal = {Biomedical Informatics and Smart Healthcare},
year = {2026},
volume = {2},
number = {1},
pages = {20-37},
doi = {10.62762/BISH.2026.470997},
url = {https://www.icck.org/article/abs/BISH.2026.470997},
abstract = {Parkinson’s disease (PD) is the second most common neurodegenerative disorder worldwide, predominantly affecting older adults. Early detection is crucial, as subtle motor and non-motor symptoms frequently overlap with other conditions, often resulting in delayed diagnosis. Many existing models rely on costly and less accessible imaging modalities such as MRI or PET scans, limiting their applicability in resource-constrained settings where only routine clinical data are available. This study develops interpretable AI models for early PD detection using structured clinical variables, incorporating feature selection techniques. Feature selection was conducted via Random Forest (RF) importance ranking combined with SelectKBest statistical scoring, retaining the most informative predictors for modeling. Five classifiers were implemented in parallel: Support Vector Machine (SVM), K-Nearest Neighbors (KNN), RF, Convolutional Neural Network (CNN), and Long Short-Term Memory (LSTM) network. Model performance was assessed using accuracy, precision, recall, F1-score, and ROC-AUC metrics. The RF model achieved the highest accuracy of 92.72\% (ROC-AUC: 0.968), with CNN and SVM showing competitive performance. LSTM exhibited balanced sensitivity and specificity, while KNN demonstrated relatively lower recall. To improve clinical interpretability, LIME was applied to each model to produce instance-level explanations, consistently highlighting tremor severity, motor impairment, cognitive scores, and age as key influential features. These results demonstrate that structured clinical variables alone can enable reliable PD detection without dependence on imaging. Integrating explainable artificial intelligence enhances transparency and supports responsible clinical adoption.},
keywords = {Parkinson’s disease, explainable artificial intelligence, machine learning, deep learning, feature selection, clinical decision support},
issn = {3068-5524},
publisher = {Institute of Central Computation and Knowledge}
}
Article Metrics
Publisher's Note
ICCK stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and Permissions
 Copyright © 2026 by the Author(s). Published by Institute of Central Computation and Knowledge. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made.
Copyright © 2026 by the Author(s). Published by Institute of Central Computation and Knowledge. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made.

Portico

